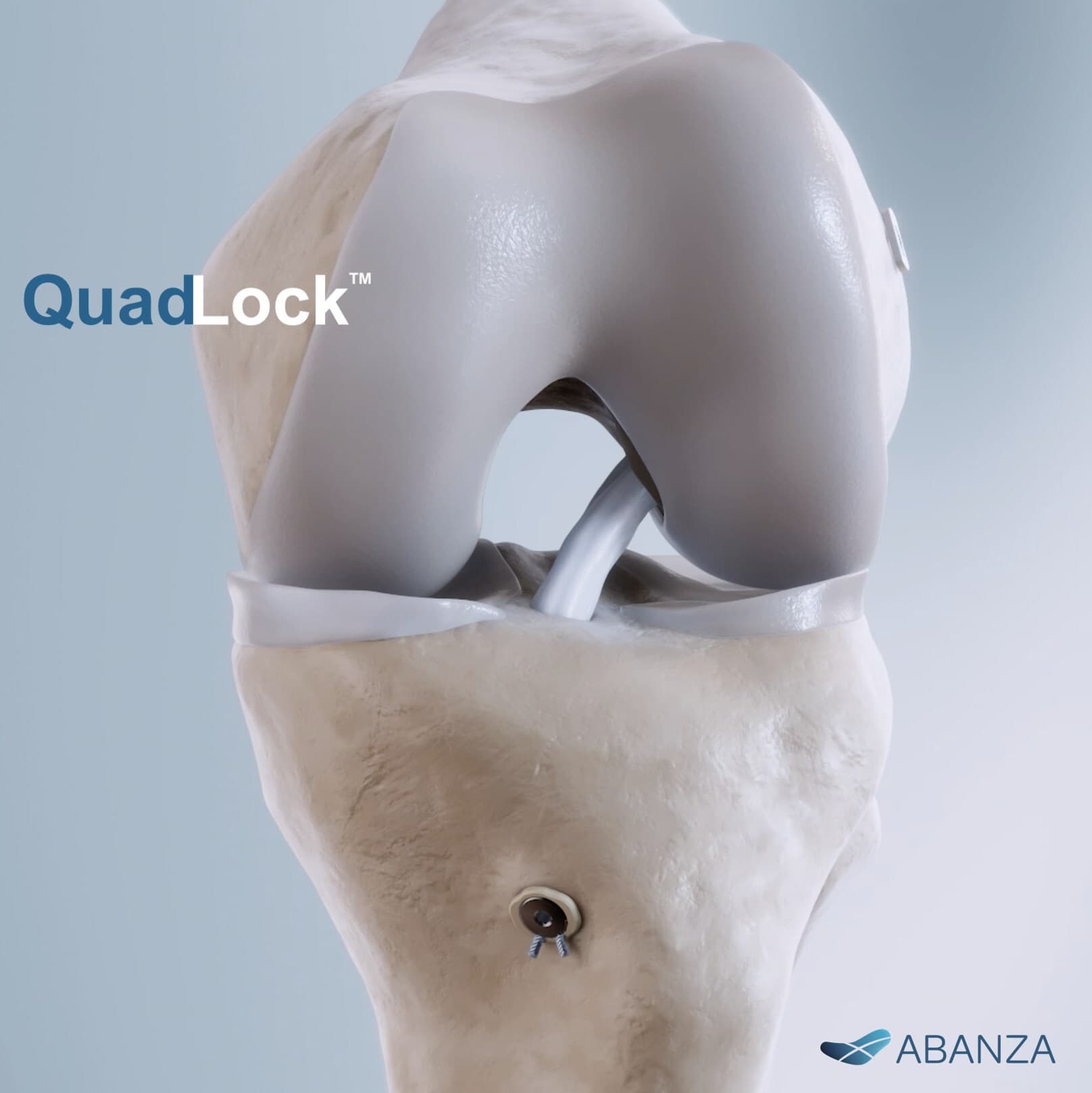
ABANZA Secures FDA 510(k) Clearance for QuadLock™ Fixation System for ACL Reconstruction
✅ FDA 510(k) Clearance Achieved — #QuadLock™ is here 🚀
We're excited to announce that ABANZA has received U.S. FDA 510(k) clearance for QuadLock™, our knotless, tension-adjustable fixation system for ACL reconstruction.
QuadLock™ is designed to help surgeons fine-tune graft tension and maintain stability across multiple ACL graft configurations, including quad tendon, hamstring, and BTB. In high-demand cyclic loading and pull-out biomechanical testing, QuadLock™ demonstrated:
- 📉 <0.5 mm cyclic displacement
- 💪 >1,000 N pullout strength
🔷 As our CEO Juan Abascal shared: "QuadLock™ reflects our focus on practical innovation—giving surgeons adjustability, control, and confidence when fixation performance matters most…"
This clearance marks an important step in our U.S. roadmap—and strengthens our broader platform strategy to advance soft-tissue repair with solutions built for a patient population that continues to evolve—from younger, high-demand athletes.
🔜 More to come as we continue building a New Era in Soft Tissue Fixation.
#FDA #510k #QuadLock #ACL #ACLSurgery #SportsMedicine #Orthopedics #MedTech #SurgicalInnovation #SoftTissueFixation #Biomechanics #ReturnToSport #InnovationInHealthcare #PatientOutcomes